
| Tweet |

Custom Search
|
|

|
||
 TM 9-8000
PART THREE
ELECTRICAL SYSTEMS AND RELATED UNITS
CHAPTER 11
BASIC PRINCIPLES OF ELECTRICITY
Section I. ELECTRICITY
that it is made up of those same basic particles having
11-1. Composition of Matter.
positive and negative electrical charges as discussed
above.
a. To understand electricity, first study matter, the
name for all material substances. Everything (solids,
c. The basic particles that make up all the
liquids, and gases) is made up of tiny particles known as
elements, and thus all the universe, are called protons,
atoms. These atoms combine in small groups of two or
electrons, and neutrons. A proton is a basic particle
more to form molecules. Air is made up of molecules.
having a single positive charge; a group of protons
These molecules are made up of atoms, and these toms
produces a positive electrical charge. An electron is a
can be further subdivided. When atoms are divided,
basic particle having a single negative charge; therefore,
smaller particles are created, some of which have
a group of electrons produces a negative electrical
positive and others, negative electrical charges. Atoms
charge. A neutron is a basic particle having no charge; a
of different materials are discussed below.
group of neutrons, therefore, would have no charge.
b. There are over 100 different basic materials in
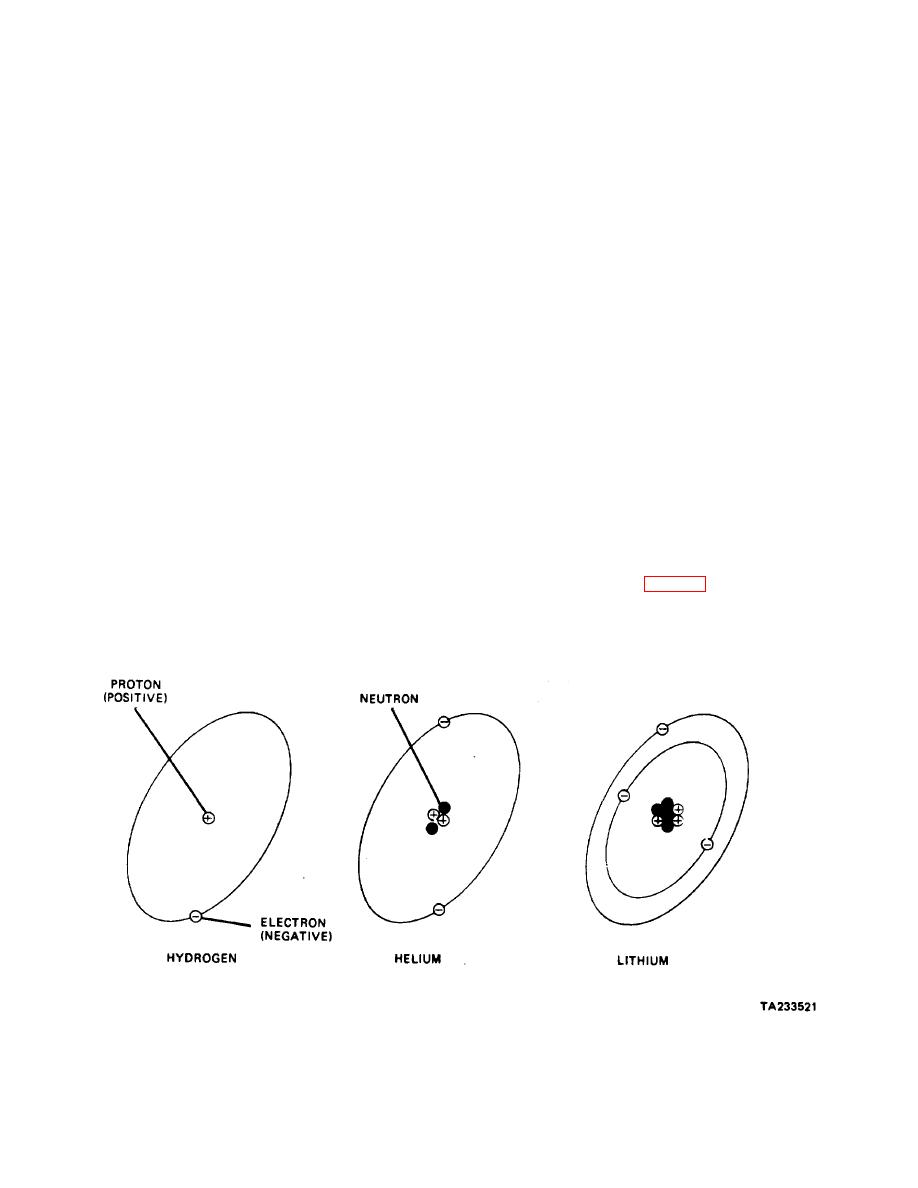
d. Examine the construction of atoms of the various
the universe. These basic materials are called elements.
elements, starting with the simplest of all, hydrogen. The
Iron is one element; copper, aluminum, oxygen,
atom of hydrogen consists of one proton, around which
hydrogen, and mercury are other elements. An element
is circling one electron (fig. 11-1). There is an attraction
gets its name from the fact that it cannot be broken down
between the two particles, because negative and positive
easily into simpler (or more elemental) substances. In
electrical charges always attract each other.
other words, more than 100 basic elements are the
building materials from which the universe is made. If
any one of these elements is studied closely, it is obvious
Figure 11-1. Composition of Matter.
11-1
|
||
 |
||